Introduction
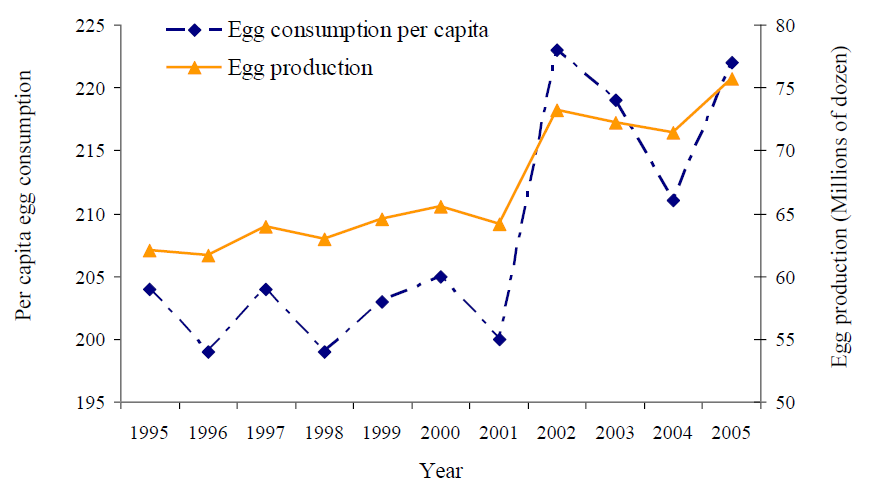
Over the past 10 years, annual per capita egg consumption in New Zealand has increased from 204 eggs (in 1995) to 222 eggs (in 2005) (Wintle, personal communication), one of the highest in the world (Williams, personal communication). Figure 1 shows the egg consumption per capita and the annual egg production in New Zealand since 1995.
Figure 1: Egg consumption per capita and egg production in New Zealand between 1995 and 2005
Using an average egg weight of 60 g, and accounting for a shell weight of approximately 11 % of the egg (MAFF, undated), it is estimated that New Zealanders consumed on average 11.9 kg of eggs in 2005. This is slightly higher than the 10.5 kg of lamb consumed per capita in the same year (Meat and Wool New Zealand Economic Service, 2006).
Due to the diversity in the potential uses of eggs and the subsequent demands of the consumer that egg quality becomes extremely difficult to define.
Defining egg quality
Kramer (1951, cited by Koelkebeck, 2006) defined quality as “the sum of characteristics of a given food item which influence the acceptability or preference for that food by the consumer”. Based on this definition, it is clear that egg quality will mean different things to different people and the consumer’s perception of quality is likely to vary depending on their intended use of the egg and their own preferences. This is clearly illustrated by a brief review of the regulatory requirements for eggs sold around the world.
Under the European Union Egg Marketing Regulations, enforced in all European Union Countries, eggs are classed as either Class A or Class B eggs (Council of the European Union, 2006; European Commission, 2003) and only eggs graded Class A can be sold for direct human consumption or retailed (Council of the European Union, 2006). The characteristics of Grade A and B eggs are detailed in Appendix 1.
Similarly, the United States Department of Agriculture (USDA) has developed three grades of eggs based on the interior quality of the egg and the appearance and condition of the egg shell (USDA Food Safety and Inspection Service, undated). U.S. Grade AA and U.S. Grade A eggs are usually retailed while U.S. Grade B eggs are usually sent for further processing. The characteristics of U.S. Grade AA, A and B eggs are detailed in Appendix 2.
In contrast, no defined grades have been developed for shell eggs sold in New Zealand and little attention has been focused on internal egg quality factors, such as yolk or albumin quality. However, the Animal Products (Specification for Products Intended for Human Consumption) Notice 2000 requires that shell eggs intended to be traded in the shell must
- be visibly clean; and
- have no cracks that are visible on candling (or equivalent) unless they have been treated by a process that destroys pathogenic organisms; and
- have no evidence of embryo development or putrefaction, and no significant blood clots; and
- not have been incubated; and
- be handled and stored under conditions that minimise condensation on the surface of the eggs.
Eggs which do not meet these minimum requirements can only be sold for human consumption if they have been pasteurised (or undergone an equivalent process) and meet the microbiological criteria specified in Standard 1.6.1.
Thus although grading systems for shell eggs may vary from country to country or region to region, all of the regardless of the grading or classification system used egg shell quality and interior quality are important factors in determining egg quality.
Egg shell quality
The vast majority of eggs sold in New Zealand are sold in their shell and a consumer’s first impression of any egg purchased is based on their perception of shell quality.
The Egg Producers Federation of New Zealand (Inc) (EPF) Code of Practice (EPF and NZFSA, 2002) lists 14 possible eggs shell defects, although these can be grouped into five main categories, defects associated with egg shell integrity, texture, shape, colour and cleanliness.
Egg shell integrity
Defects considered under the category of egg shell integrity include gross cracks, hairline cracks, star cracks and thin shelled or shell-less eggs. As cracked eggs cannot be made available for retail sale in New Zealand (Food Standards Australia New Zealand, 2006), high numbers of cracked eggs will have a negative impact on the profitability of any egg producer.
One of the most obvious reasons for egg shell cracks (including gross cracks, hairline cracks and star cracks) is mechanical damage caused either by the birds themselves or as a result of poor management practices, such as infrequent collection of eggs, rough handling and poor design and / or maintenance of the cage floor.
Egg shell strength ultimately affects the soundness of the shell, with weaker shelled eggs more prone to cracks and breakages and subsequently microbial contamination. Shell strength can be affected by a wide range of factors including:
Egg size: Smaller eggs have stronger shells than larger ones, as hens have a finite capacity to deposit calcium in the shell and as a result, the same amount of calcium is spread over a larger area (Butcher and Miles, 2003a).
Age of bird: Older birds tend to lay bigger eggs and have a higher egg output, which impacts on shell strength as described above (Butcher and Miles, 2003a). Very young birds with immature shell glands may produce shell-less eggs or eggs with very thin shells. Delaying the onset of sexual maturity by one to two weeks will prevent this (Coutts and Wilson, 1990).
Stress: A single stress or disturbance to a flock of laying hens can be enough to de-synchronise the process of egg formation for several days, during which time, a number of different egg quality faults may be seen (MAFF, undated). For example;
- Any factor which results in oviposition prior to completion of shell deposition will result in soft or thin-shelled eggs. Activities which create disturbances in and around the layer shed should be minimised (Coutts and Wilson, 1990).
- If an egg is retained in the shell gland, any subsequent egg laid may spend less time than normal in the shell gland, resulting in insufficient shell deposition and a soft shelled or shell-less egg (MAFF, undated).
Solomon (1991) noted that once an imperfect mammillary layer has formed (as a result of stress experienced before the egg reaches the shell gland), subsequent layers are disorganised and thin or soft shelled eggs are a common phenomenon after stress.
Elevated environmental temperature: High (above 25oC) environmental or shed temperatures may affect the feed (and therefore calcium) intake of the bird, thus resulting in a decreased availability of calcium for shell deposition. As well as decreasing feed intake, laying hens will try to overcome heat stress by panting (Koelkebeck, 1999). However, this causes a decrease in the amount of carbon dioxide (CO2) in the hens’ blood, a condition known as respiratory alkalosis (Koelkebeck, 1999). As egg shells are made up of 95 % calcium carbonate (CaCO3), this decrease in blood CO2 levels, combined with an increase in blood pH and a subsequent decrease in Ca2+ ions for shell formation leads to an increase in the number of thin or soft shelled eggs produced. Arima et al. (1976, cited by Jones, 2006) found that the egg quality of older hens was more severely affected by increased temperature than younger hens.
Nutrition and water quality: The provision of adequate dietary minerals and vitamins is essential for good eggshell quality. Similarly, as water quality varies from country to country and region, the role of drinking water in mineral and trace element supply should not be overlooked.
Calcium and phosphorous are essential macro minerals with calcium forming a significant component of the shell and phosphorous playing an important role in skeletal calcium deposition and subsequent availability of calcium for egg shell formation during the dark period (Boorman et al., 1989).
Coetzee (2002) investigated the effect of calcium levels in drinking water on shell integrity in South African laying hens and demonstrated that birds supplied an additional 200 mg of calcium per litre of drinking water laid eggs with a mean shell strength of 42.6 ± 9.0. This was in comparison to those receiving un-supplemented water, whose eggs had a mean shell strength of 38.9 ± 7.0.
However, the feeding of calcium levels above the requirement of the bird for production has not been shown to improve shell quality (Kershavarz and Nakajima, 1993). Indeed, feeding hens high levels of calcium may interfere with the availability of other minerals (NRC, 1994) and can have a negative impact on the ability of the bird to utilise calcium, particularly if calcium levels in the diet are subsequently decreased. This is particularly true in young pullets. Wideman and Lent (1991, cited by Summers, undated) also reported that high levels of calcium can cause latent kidney damage, which has a long term impact on bird survivability.
Similarly, feeding high levels of dietary phosphorous have also been shown to have a negative effect on eggshell quality (Kershavarz and Austic, 1990, cited by Jones, 2006; Harms, 1982a, cited by Boorman, et al., 1989; Harms, 1982b, cited by Boorman, et al., 1989; Taylor, 1965, cited by Boorman, et al., 1989; Arscott, et al., 1962, cited by Boorman, et al., 1989). Although Miles and Harms (1982, cited by Boorman et al., 1989) showed a clear linear negative correlation between specific gravity and plasma phosphate, it remains unclear whether excess plasma phosphorous interferes with the mobilization of skeletal phosphorous reserves or the shelling process itself (Ahmad and Balander, 2004; Boorman et al., 1989) or if elevated levels of phosphorous increases calcium excretion (Keshavarz and Austic, 1990, cited by Jones, 2006).
Keshavarz and Austic (1990, cited by Jones, 2006) also examined the interaction of phosphorus and chloride and the role of chloride in egg shell integrity. As with phosphorous, elevated dietary levels of chloride resulted in decreased eggshell quality and lower levels of blood acid-base indicators. This is supported by the work of Balnave et al. (1989) who observed an increase in shell defects with no changes in egg production, egg weight, feed or water intake, blood-acid base levels and electrolyte levels for birds provided with 2000 mg of sodium chloride (NaCl) per litre drinking water. This was in comparison to those hens provided with water with 600 mg NaCl per litre. Birds receiving 2000 mg of NaCl per litre drinking water also had an increased incidence of shell-less eggs. Egg shell defects persisted after the sodium chloride was removed from the drinking water. In contrast, Hess and Britton (1989; cited by Jones, 2006) fed hens diets low in chloride and found virtually no effect on shell quality.
Trace mineral nutrition is a complex area of animal nutrition, and a wide range of interactions and antagonisms can result in poor absorption or utilisation of minerals essential for shell formation. It should also be noted that not all trace mineral sources are equally available, and consideration should be given to this aspect of premix formulation. Burley and Vadehra (1989, cited by Miles, 2001) and Mas and Arola (1985, cited by Miles, 2001) reported varying amounts of zinc, copper, iron and manganese in the shell and its associated membranes. It is clear, therefore, that provision of adequate levels of these minerals, which are key components of the shell matrix and play an essential role as co-enzymes, is essential if shell integrity is to be maintained.
Vitamin D also plays an important role in the proper utilisation of calcium and phosphorous and sufficient amounts of this vitamin should be included in the feed.
Finally, care should be paid to the mixing of the diet. Thorough mixing of the feed is essential if each bird in the flock is to receive a similar amount of any given nutrient. This is particularly true for layer hen diets which frequently contain raw materials with a wide range of different densities.
Mycotoxicosis: Jewers (1990) reported that thin rubbery shells which break more readily than normal have been observed during field outbreaks of ochratoxicosis. Similarly, in an outbreak of T-2 toxicosis, egg breakages increased from a normal 3 % to 15 % with a further 18 % of eggs broken in transit to customers (Jewers, 1990). Zaghini et al. (2005) reported that birds fed diets containing 2.5 ppm of aflatoxin B1 had lower egg shell weights than those fed the control diet or diets supplemented with mannanoligosaccharides.
Genetics: Clunies et al. (1992, cited by Jones, 2006) found that hens laying thick-shelled eggs retained more dietary calcium than those laying thin-shelled eggs. Although there was no difference in egg production between thick and thin shell layers, both egg and shell weight were greater for the thick shelled eggs.
Disease: Infectious bronchitis (IB), a viral disease caused by a coronavirus which attacks the mucus membranes of the respiratory and reproductive tracts (Jones, 2006; Butcher and Miles, 2003a; Cavanagh and Naqi, 2003), may result in egg defects. These include pale shelled eggs, and eggs with poor shell structure and integrity (Jones, 2006; Beyer, 2005; Butcher and Miles, 2003a; Cavanagh and Naqi, 2003). Similarly, birds affected by egg drop syndrome (EDS), caused by an adenovirus, initially produce pale eggs, quickly followed by thin-, soft-shelled or shell-less eggs (McFerran and Adair, 2003).
Texture
Rough or “sandpaper” shells, pimples, pinholes and mottled or glassy-shells are all egg shell defects associated with egg shell texture. These defects are frequently a result of bird age, but may also be caused by other factors (Coutts and Wilson, 1990; MAFF, undated).
Disease: Certain diseases such as IB, infectious laryngotracheitis (ILT) (Beyer, 2005; Coutts and Wilson, 1990) and avian encephalomyelitis (AE) (Coutts and Wilson, 1990) have been implicated in the production of rough or “sandpaper” eggshells.
Mycotoxicosis: As discussed above, ochratoxicosis may result in rubbery-shelled eggs (Jewers, 1990).
Genetics: The production of eggs with calcium deposits on the shell (or pimples) is thought to be hereditary (Butcher and Miles, 2003a; Coutts and Wilson, 1990).
Management: Overcrowding of birds, changes in the lighting programme, poor shed ventilation and inadequate water supply can contribute to increased incidence of shell defects associated with egg texture (Coutts and Wilson, 1990).
Shape
Misshapen eggs have a shape which differs from the smooth normal shape (for example, flat sided eggs and body checked eggs). This can be caused by a number of factors
Age of bird: As with shell soundness, young birds with immature shell glands may produce misshapen eggs (Coutts and Wilson, 1990).
Stress: Body checked eggs, marked by grooves and ridges, occur when the shell of the egg breaks in the shell gland, during the formation process (i.e. 10 – 14 hours before the egg is laid). Although the damage can be partly repaired, a bulge forms around the egg (Solomon, 1991; Coutts and Wilson, 1990; MAFF, undated). Flat sided eggs occur where two eggs are in the shell gland at the same time (Solomon, 1991; MAFF, undated). Both defects may be caused by overcrowding, frights or other disturbances and poor lighting patterns (Koelkebeck, 1999; Coutts and Wilson, 1990). Jones (2006) stated that proper handling can reduce the incidence of body checks.
Disease: As the albumen of the egg and surrounding membranes provides the structure on which the egg shell is deposited, if the albumen quality is very poor, there it not sound foundation on which to build the shell (MAFF, undated). As a result, those diseases which result in poor albumen quality often cause an increase in the number of misshapen eggs. Examples of these are IB (Jones, 2006; Butcher and Miles, 2003a; Cavanagh and Naqi, 2003), EDS (McFerran and Adair, 2003; Coutts and Wilson, 1990) and certain strains of Newcastle Disease (NCD) or avian influenza (AI) (Butcher and Miles, 2003a; Coutts and Wilson, 1990).
Colour
The colour of an egg shell is determined primarily by the genetics of the hen, with white feathered hens laying white eggs and brown feathered hens laying brown eggs (Jacob et al., 2000).
During the process of egg shell formation in brown egg layers, the epithelial cells lining the surface of the shell gland synthesise and accumulate pigments, such as biliverdin-IX, its zinc chelate and protoporphyrin-IX (Butcher and Miles, 2003b). In the final three to four hours of shell formation these pigments are transferred to the viscous, protein rich cuticle. The quantity of pigment in the cuticle which determines the colour of the egg (Butcher and Miles, 2003b). As the cuticle is deposited onto the eggshell at the same time that shell deposition reaches a plateau (approximately 90 minutes prior to oviposition), pigment distribution is not uniform throughout the shell, with very little pigment contained in the shell itself (Butcher and Miles, 2003b). Thus, any factor which causes a disruption, either in the ability of the epithelial cells to synthesise pigment or in the deposition of the cuticle, will affect the colour of the egg shell. These factors include:
Stress: Epinephrine, a stress hormone, will cause a delay in oviposition and cessation of shell gland cuticle formation, which can cause pale shelled eggs to be produced. Stressors may include, amongst others, high cage density, loud noise and handling (Butcher and Miles, 2003b).
Age of bird: As birds age increases, the intensity of pigment decreases. This may be due to decreasing production of pigment or increased surface area over which available pigment is distributed (Butcher and Miles, 2003b).
Chemotherapeutic agents: Certain drugs have been shown to affect egg shell colour. For example, nicarbazin (an anticoccidial drug) fed at a level of 5 mg per day can result in the production of pale eggs within 24 hours, while higher doses can lead to complete depigmentation (Butcher and Miles, 2003b). Chlortetracycline (600 – 800 ppm) may also result in yellow egg shells (Beyer, 2005; Hendrix Genetics, undated).
Disease: Viruses, which affect the mucus membranes of the respiratory and reproductive tract, such as NCD and IB, not only cause a decrease in egg production, but also cause the shell to become abnormally thin and pale (Beyer, 2005; Butcher and Miles, 2003b).
Cleanliness
Cleanliness is probably the easiest aspect of egg shell quality to control, and good management plays an important role. Most eggs are clean when laid and subsequently become contaminated with faecal material or other contaminants. Defects listed in the EPF Code of Practice (EPF and NZFSA, 2002), which fall into this category, include cage marks, stained eggs and fly marks. Although fungus or mildew on shells is listed as a defect, it is only likely to occur in poor conditions (Coutts and Wilson, 1990).
Management: Good management practices will help reduce the number of dirty eggs. These practices include frequent collection of eggs, as well as regular replacement of litter material in nest boxes or regular maintenance and cleaning of cage floors and roll out trays. Whilst less common, fly stains, water stains and grease or oil stains may occur, and can be prevented by good shed and equipment maintenance or management (Coutts and Wilson, 1990).
Nutrition and / or bird health: Any factor which causes diarrhoea in the birds, (for example high dietary salt levels), will also result in an increase in the number of dirty eggs collected. Blood smears on eggs can be minimised by good pullet management, including weight for age, lighting and beak trimming if necessary (Coutts and Wilson, 1990).
Internal egg quality
Unlike external (shell) quality, internal quality of the egg begins to decline as soon as the egg is laid. Thus although factors associated with the management and nutrition of the hen do play a role in internal egg quality, egg handling and storage practices do have a significant impact on the quality of the egg reaching the consumer. Similarly, although the shell provides a unique “package” for the distribution of the egg contents to the consumer, it is in fact the internal quality of the egg that is most important to the consumer. These aspects of internal quality are significantly more difficult to observe or evaluate in the intact egg, even with the use of candling.
In addition to the, obvious, nutritional quality of the egg, internal egg quality is extremely important because of its many functional and aesthetic properties. For example, eggs are used as thickening agents in custards and puddings, egg whites are used as smoothing agents to give icings a desirable texture and egg yolks add colour and richness to food (Koelkebeck, 1999).
In recent years, much attention has been focused on increasing the omega 3:6 ratio and vitamin content of eggs, principally through manipulation of the diet. However, although these “enhancements” further complicate the issue of egg quality, it is beyond the scope of this review to discuss these further.
The EPF Code of Practice (EPF and NZFSA, 2002) lists a total of nine internal defects and these can be broadly categorised into three groups; namely: defects affecting yolk quality, defects affecting albumin quality and defects affecting overall quality.
Yolk quality
Yolk quality is determined by the colour, texture, firmness and smell of the yolk (Jacob et al., 2000).
Yolk colour: Although yolk colour is a key factor in any consumer survey relating to egg quality (Jacob et al., 2000), consumer preferences for yolk colour are highly subjective and vary widely from country to country. In general, New Zealand consumers prefer a yolk colour between 11 and 13 on the Roche (now DSM) Yolk Colour Fan (Sutherland, personal communication).
The primary determinant of yolk colour is the xanthophyll (plant pigment) content of the diet consumed. It is possible to manipulate the yolk colour of eggs by the addition of natural or synthetic xanthophylls to layer hen feeds. This ability to readily manipulate egg yolk colour can be an advantage in meeting market demands. However, the ease with which yolk colour can be manipulated can lead to unwanted colour changes. For example, the inclusion of higher than recommended levels or incorrect ratios of pigments can lead to orange-red yolks (Coutts and Wilson, 1990). Similarly, diphenyl-para-phenylenediamine (DPPD), an antioxidant, has been reported to cause excessive deposition of pigments in the egg yolk (Coutts and Wilson, 1990). The inclusion of more than 5 % cottonseed meal in a layer diet will result in olive or salmon coloured yolks (Beyer, 2005), while the inclusion of certain weeds or weed seeds may results in green yolks (Beyer, 2005; Coutts and Wilson, 1990). Similarly, inadvertent omission of xanthophylls from the diet will lead to pale yolks. Both inadequate mixing of the diet as as well as excessive mixing of the diet will also result in a heterogeneous feed, and subsequent variation in the amount of xanthophylls consumed by each hen in the flock, This will result in egg yolk colour not being uniform throughout the flock.
Pale yolks can result from any factor which alters or prevents the absorption of pigments from the diet or the deposition of these pigments in the yolk. These factors could include;
- worms (Coutts and Wilson, 1990)
- any factor which inhibits liver function, subsequent lipids metabolism and deposition of pigment in the yolk. For example, mycotoxicosis caused by aflatoxin B1 (Zaghini et al., 2005) and
- coccidiosis, although this is rare in adult hens.
Mottled yolks ( with many pale spots and blotches which vary in colour size and shape), occur when the contents of the albumen and yolk mix as a result of degeneration and increase permeability of the vitelline membrane (Jacob et al., 2000). Factors affecting mottling were reviewed in detail by Cunningham and Sanford (1974).Dietary factors which may cause mottled yolks include;
- the presence of nicarbazin (an anticoccidal agent) in the feed has shown by numerous authors to cause mottling (Jones et al., 1990; Cunningham and Sanford, 1974)
- worming drugs such as phenothiazine (Coutts and Wilson, 1990), dibutyltin dialaurate (Jacob et al., 2000; Coutts and Wilson, 1990; Berry et al., 1968, cited by Cunningham and Sanford, 1974) and Piperazine (Jacob et al., 2000; Coutts and Wilson, 1990). However, Berry et al. (1968, cited by Cunningham and Sanford, 1974) did not observe yolk defects when Piperazine was fed at the manufacturer’s recommendations. Similarly, they only observed defects when dibutyltin dialaurate was fed at the recommended level but over a much longer period.
- gossypol from cotton seed meal (Jacob et al., 2000; Berry et al., 1968, cited by Cunningham and Sanford, 1974)
- certain antioxidants such as gallic acid (from grapes, tea and oak bark) and tannic acid (Coutts and Wilson, 1990), or tannins from grains such as sorghum (Jacob et al., 2000)
- calcium deficient diets (Jacob et al., 2000; McCready et al., 1972, cited by Cunningham and Sanford, 1974)
Storage time and temperature has also been shown to affect the degree of egg yolk mottling (Jacob et al., 2000; Coutts and Wilson, 1990). Jones (2006) stated that as the internal temperature of the egg increases above 7oC, the protein structures of the thick albumen and vitelline membrane breakdown faster. As the membrane degenerates during storage, water enters the yolk causing mottling and after prolonged storage, albumen proteins also enter the yolk increasing the severity of mottling (Jacob et al., 2000). In order to reduce the rate of breakdown of the vitelline membrane, eggs should be collected regularly, reducing the time they are exposed to higher environmental temperatures and contaminants, and stored at temperatures of 7 – 13oC and humidity of 50 – 60 %.
In their review, Cunningham and Sanford (1974) also identified hen age, oil coating of eggs and movement of eggs as possible factors affecting mottling of eggs.
Yolk firmness: The yolk of a freshly laid egg is round and firm (Jacob et al., 2000). However, as the egg ages and the vitelline membrane degenerates, water from the albumen moves into the yolk and gives the yolk a flattened shape.
Yolk texture: Rubbery yolks may be caused by severe chilling or freezing of intact eggs, the consumption of crude cottonseed oil or the seeds of some weeds (Jacob et al., 2000)
Albumin quality
Albumin quality is related to the consistency, appearance and the functional properties.
Consistency: Albumin quality is measured in terms of Haugh Units (HU) calculated from the height of the albumin and the weight of the egg (Coutts and Wilson, 1990; MAFF, undated). A minimum measurement in HU for eggs reaching the consumer is 60 (Craig, personal communication). However most eggs leaving the farm should be between 75 and 85 HU (Coutts and Wilson, 1990). Albumin consistency is influenced by:
Age of the hen: HU will decrease with increasing bird age value, with HU decreasing by around 1.5 to 2 units (Coutts and Wilson, 1990) for each month in lay. Doyon et al. (1986, cited by Jones, 2006) stated that HU decreases at a fairly constant rate of 0.0458 units per day of lay as the hen ages. Craig (personal communication) noted that in an ideal situation, HU should be on average 102 at 20 weeks of age, falling to an average of 74 HU by 78 weeks of age.
Genetics: Strain of bird has also been shown to play a role in albumin consistency, with some strains consistently producing eggs with thin albumin. Curtis et al. (1985, cited by Jones, 2006) reported that brown egg layers produced eggs with higher HU, while other authors (Williams, 1992, cited by Jones, 2006; Hill, 1981, cited by Jones, 2006) reported that HU values were more variable within the brown egg layers compared with those that lay white eggs. High producing birds tend to lay eggs with relatively lower amounts of thick albumin and, although this can be influenced by selective breeding, egg numbers are usually considered more important.
Age and storage of the egg: As the egg ages and carbon dioxide (CO2) is lost through the shell, the contents of the egg become more alkaline, causing the albumin to become transparent and increasingly watery (MAFF, undated). At higher temperatures, loss of CO2 is faster and the albumin quality deteriorates faster (MAFF, undated). Decreasing shed temperatures in the hotter months, combined with regular collection of eggs will help to reduce deterioration of the albumin before collection (Jones, 2006; Coutts and Wilson, 1990).
Eggs stored at ambient temperatures and humidity lower than 70 % will lose 10 – 15 HU in a few days from point of lay. By 35 days, these eggs will lose up to 30 HU (Craig, personal communication). Storage of eggs at temperatures of 7 – 13oC and a humidity of 50 – 60 % (as discussed under mottling), will reduce the rate of degeneration of thick albumen proteins and, consequently, egg albumin quality will be maintained for longer (Jones, 2006).
Oiling of eggs can also help to reduce CO2 losses and thus help maintain internal egg quality (Beyer, 2005; Koelkebeck, 1999; Coutts and Wilson, 1990) but is not a substitute for cool storage (Jacob et al., 2000).
Vanadium: Henry and Miles (2001) reviewed the effects of vanadium on poultry performance. They noted that poorer albumin quality has been reported from laying hens consuming as little as 6 ppm (Sell et al., 1982, cited by Henry and Miles, 2001). Later reports (Sell et al., 1986, cited by Henry and Miles, 2001) showed that the interior quality of eggs decreased in two strains of laying hens fed 3 or 6 ppm added vanadium. Duyck et al. (1990, cited by Henry and Miles, 2001) fed laying hens 10 ppm of vanadium for 30 days. HU from these hens averaged 71 HU after one day of storage (62 oF (16.6 oC) and 60 % Relative Humidity) and 64 after seven days of storage. This was in contrast to the average of 82 and 74 HU after one and seven days storage respectively, observed for hens fed the control diet. Henry and Miles (2001) reported that the negative effects of vanadium may be overcome by feeding cottonseed meal, ascorbic acid, vitamin E or carotene, although this is dose dependant.
Diseases: Diseases such as certain strains of EDS, IB (Jacob et al., 2000; Coutts and Wilson, 1990), NCD and ILT (Jacob et al., 2000) can all cause a decrease in albumin consistency.
Appearance: Normal albumin is transparent, with a slightly yellow green colour. Discolouration of the albumin may occur if the eggs are stored for an extended time period in poor conditions, with the albumin becoming much yellower (Coutts and Wilson, 1990). Cyclopropene fatty acids from cottonseed meal (Beyer, 2005; Coutts and Wilson, 1990) and the certain weed seeds (Coutts and Wilson, 1990) can cause albumin to turn pink after storage. Green whites are caused by excesses of riboflavin (vitamin B2) in the diet (Coutts and Wilson, 1990). Cloudy whites may be caused by the oiling of eggs within six hours of lay (Beyer, 2005).
Overall quality
Blood spots: Blood spots may vary from indistinguishable spots on the surface of the yolk to heavy contamination throughout the yolk (Coutts and Wilson, 1990). Although blood spots are normally closely associated with the yolk (MAFF, undated), occasionally blood may be diffused through the albumin (Coutts and Wilson, 1990). Blood spots occur when small blood vessels in the ovary rupture when the yolk is released (MAFF, undated).
Vitamin deficiency: Vitamin K plays an important role in blood clotting. Vitamin K deficiency can result in an increased occurrence of blood spots (Bains, 1999)
Genetics: Some strains of birds appear to be predisposed to blood spots (Coutts and Wilson, 1990) although the incidence is low (Beyer, 2005).
Disease: Avian encephalomyelitis has been reported as a cause of blood spots (Coutts and Wilson, 1990).
Mycotoxicosis: Jewers (1990) reported an increase in blood spots from “essentially 0 to 3 %” in birds affected with T-2 toxicosis. Bains (1999) suggested that mycotoxicosis may reduce vitamin K absorption and this may explain the elevated incidence of blood spots in hens affected by T-2 toxicosis.
Meat spots: These are usually associated with the albumin (MAFF, undated) rather than the yolk and often consist of small pieces of body tissue (Coutts and Wilson, 1990; MAFF, undated). However, some may consist of partially broken down blood spots (Coutts and Wilson, 1990) or pigments. The occurrence of blood spots varies with strain of bird, increases with age of bird and is reported to be higher in brown egg layers (Coutts and Wilson, 1990; MAFF, undated).
Bacterial or fungal contamination: Solomon (1991) suggested that while pores on the surface of the egg do represent possible ports of entry for bacteria, particularly as the cuticle hardens just after oviposition, these are of secondary importance to the structural defects that may occur. Structural defects, because of their magnitude, offer a much more likely route for bacteria to enter the egg contents.
Bacterial and fungal contamination of eggs usually results in black, red or green rot. The egg looks and smells putrid when broken out of the shell (Beyer, 2005; Coutts and Wilson, 1990).
Bacterial and fungal contamination of eggs, resulting from faecal contamination of the egg, can be prevented by good management practices, including regular replacement of nesting materials or good cage maintenance as appropriate (Beyer, 2005; Coutts and Wilson, 1990). Bacterial contamination of the egg contents may also occur as a result of an infection in the oviduct of the hen, and any affected hens should be culled (Coutts and Wilson, 1990).
Proper handling and storage of eggs following collection will minimise the opportunity for bacterial or fungal contamination. However, improper washing procedures, high storage temperatures and humidity will increase the incidence of bacterial of fungal contamination (Coutts and Wilson, 1990).
Careful attention should be paid to feed source, as Salmonella spp. can be transmitted through the feed. New Zealand is fortunate to have considerably lower (almost insignificant) levels of Salmonella contamination of eggs compared to that observed in other countries around the world (Brooks, personal communication).
Roundworms in eggs: Coutts and Wilson (1990) reported that where roundworm infestation of the intestinal tract occurs, worms may migrate from the cloaca into the oviduct and become enclosed in the egg. This can be prevented by good flock management.
Off odours / flavours: Although off odours and flavours are rare if eggs are stored correctly (Coutts and Wilson, 1990), eggs readily absorb strong odours or flavours. Storage of eggs in close proximity to fish oils and meals, sour milk, strongly scented or decaying fruit and vegetables, mould, disinfectants and kerosene is likely to result in the development of off odours or flavours (Coutts and Wilson, 1990). However, eggs that have been oiled are less likely to absorb foreign odours (Coutts and Wilson, 1990). Old eggs and eggs stored at high temperatures are more likely to exhibit off odours or flavours.
Other causes of off odours or flavours include strongly flavoured feed ingredients such as fish meal or fish oil, some vegetables (including onions, turnips and excessive amounts of cabbage) and rapeseed or canola (Coutts and Wilson, 1990).
Conclusions
The consumer seldom sees many of the egg defects detailed above, as eggs are graded and most defects are normally removed prior to retail. Likewise, New Zealand has a unique poultry health status and the incidence of disease, and consequently egg defects resulting from disease, is low.
Producer choice of bird strain, both internationally and subsequently locally, is somewhat limited. However, the international breeding companies are very aware of the market requirements for egg quality and do include these traits in their selection programmes, where possible.
Assuming adequate nutrition, the use of suitable raw materials, proper feed hygiene and a lack of contamination of the birds’ diet with foreign matter and / or objects, there is little effect of nutrition on egg defects.
Flock, shed and feed management, egg storage and egg handling remain the three most important factors in determining the incidence of egg quality defects in New Zealand laying flocks.
Eggs remain a cheap, nutrition and popular food for New Zealand consumers.
Appendix 1: Characteristics of Grade A and B eggs as prescribed by the European Commission (2003)
Characteristics of Grade A eggs
- Grade A eggs shall have the following minimum characteristics:
- shell and cuticle: normal, clean, undamaged
- air space: height not exceeding 6 mm, stationary;
however, for eggs to be marketed as ‘extra’ it may not exceed 4 mm; - white: clear, limpid, of gelatinous consistency, free of extraneous matter of any kind;
- yolk: visible on candling as a shadow only without clearly discernible outline, not moving appreciably away from the centre of the egg on rotation, free of extraneous matter of any kind;
- germ cell: imperceptible development;
- odour: free of extraneous odours
- Grade A eggs shall not be washed, or cleaned by any other means, before or after grading.
Accordingly, eggs washed in accordance with Article 6(4) of Regulation (EEC) No 1907/90, even where they fulfil the criteria applicable to grade A eggs, may not be marketed as grade A eggs and must be marked ‘washed eggs’.
- Grade A eggs shall not be treated for preservation or chilled in premises or plants where the temperature is artificially maintained at less than 5 ºC. However, eggs which have been kept at a temperature below 5 ºC during transport of not more than 24 hours or on retail premises or in annexes thereto shall not be considered as chilled, provided the quantity stored in these annexes does not exceed the requirements for three days of retail sale on the premises in question.
Accordingly, eggs chilled in accordance with Article 6(5) of Regulation (EEC) No 1907/90 may not be marketed as grade A eggs, even where they fulfil the criteria applicable to grade A eggs. They shall be marketed as ‘chilled eggs’.
Characteristics of Grade B eggs
Grade B eggs shall be eggs which do not meet the requirements applicable to eggs in grade A. They may be passed only to food industry undertakings approved in accordance with Article 6 of Directive 89/437/EEC or to non-food industry undertakings.
Appendix 2: Characteristics of U.S. Grade AA, A and B eggs as prescribed by the USDA Food Inspection Service (undated)
U.S. Grade AA eggs have whites that are thick and firm; yolks that are high, round, and practically free from defects; and clean, unbroken shells. Grade AA and Grade A eggs are best for frying and poaching where appearance is important, and for any other purpose.
U.S. Grade A eggs have characteristics of Grade AA eggs except that the whites are “reasonably” firm. This is the quality most often sold in stores.
U.S. Grade B eggs have whites that may be thinner and yolks that may be wider and flatter than eggs of higher grades. The shells must be unbroken, but may show slight stains. This quality is seldom found in retail stores because they are usually used to make liquid, frozen, and dried egg products.
- References available on request
- This paper was first published in the Proceedings of the New Zealand Poultry Industry Conference, 2006.